Ph.D./Associate Professor Takeyuki Kikuchi Department of Materials Science and Chemistry, Graduate School of Engineering, University of Hyogo
Graduated from Department of Applied Chemistry, Faculty of Engineering, Okayama University in March 1994
Completed Master’s Program in Department of Applied Chemistry, Graduate School of Engineering, Okayama University in March 1996
Completed Doctor’s Program in Department of Applied Chemistry, Graduate School of Natural Science and Technology, Okayama University in March 1999
Worked as an assistant at the Himeji Institute of Technology before taking current position as associate professor at University of Hyogo.
Associate professor Takeyuki Kikuchi of the University of Hyogo Graduate School began his career with research on high-temperature superconductivity, and he gradually made his way to research on ferrites. He is creating materials with completely new functions by replacement at the molecular level. Several years ago, he successfully produced Sr-Co-Fe-based Z-type hexagonal ferrites. This material was reported to exhibit multiferroic properties. If used as a material in electronic components, it could be an ideal material capable of controlling both the electric and magnetic fields. This has led to ever-growing expectations for future practical applications of this material.
Research on High Coercive Force Ferrites Based on Inorganic Material Synthesis Technology
The technology for synthesizing inorganic materials is a specialized field. Organic refers to chemical compounds that contain carbon, and all other compounds are referred to as inorganic. In particular, Dr. Kikuchi is engaged in the synthesis of metal oxides, which bond metal and oxygen. Recently, attention has been focused on research on magnetic materials known as ferrites having iron oxide as its main component. Among the various types of ferrites, he has selected to focus on so-called hexagonal ferrites, which are ferrites having a hexagonal crystal structure. He is conducting his research on topics such as enhancing the performance of permanent magnets.
The best permanent magnets are magnets capable of stable and continuous generation of a strong magnetic field and have a small volume. Magnetization is dependent on the chemical composition and crystal structure, and coercive force is dependent on the chemical composition and crystal structure and the microstructure. In other words, controlling the microstructure enables improved coercive force for enhanced functional performance.
Hexagonal ferrites are metal oxides composed of iron (Fe), divalent metal, and an element“A.”Some well-known elements contained in A include barium (Ba), lead (Pb), and strontium (Sr). Improvements in synthesis processes have enabled synthesis at lower temperatures for successful miniaturization of ferrite particles. This process changes the properties so that they exhibit a strong coercive force.
Possibilities Opened by Polymerizable Complex Method, Where Ba and Sr Are Replaced at the Molecular Level
In (Ba,Sr)-Co-Fe-based Z-type ferrites, higher ratios of Sr result in a more difficult synthesis process. In the typical synthesis method, the material is mechanically pulverized, and then heat is applied to synthesize the desired product. However, it is difficult to achieve pulverization of solid raw materials at the submicron level and lower, and so mixing is not possible below these levels. Heat is applied to the pulverized material to diffuse it, and then chemical reactions are used to mix it. In previous studies, there are no examples of completely replacing Ba with Sr, and although some studies mentioned this possibility, no successful experiments of pure syntheses were found.
Associate professor Kikuchi turned his attention to the synthesis method at the molecular level. The raw materials were dissolved in water to make an aqueous solution, and this was stirred to achieve a uniform mixture. Next, the complex was formed, and a high molecular weight was achieved by polymerization between complexes. This mixed state was solidified and sintered. Even for metal oxides, dissolving is possible using an acid solution. Converting to a liquid solution enables a well-blended mixture of the composite elements and higher reactivity. This synthesis method is called the polymerizable complex method, and it is a method that has been widely known in the synthesis of high-Tc superconductor materials. Associate professor Kikuchi used this method successfully to synthesize pure Z-type ferrite with Sr completely substituted for Ba.
The Z-type ferrites produced in this way are expected to be developed for practical applications as materials for electronic components used in radio frequency regions. Many conventional ferrites are so-called spinel ferrites, but changing these to hexagonal ferrites will enable a higher magnetic permeability (magnetic response to an applied magnetic field) at radio frequencies. Associate professor Kikuchi explains, “Although the issue of the high sintering temperature remains, we have gained momentum for developing these materials for practical applications as materials for electronic components.”
Polymerizable Complex Method
This is one method of achieving precise chemical synthesis. The component metal salts are dissolved in water, citric acid is added to create a “complex,” and then a glycol or other substances are added to form an ester bond with the citric acid, and heating this yields a polymerization reaction for producing a gel. Here, the desired elements are mixed inside the gel, but because some organic substances still remain, this gel is heated to burn them off. Finally, heat is used, but compared to the conventional synthesis method, synthesis is possible at lower temperatures, and the compound becomes a liquid and gel during the process, and so it is possible to make it into thin films or extend it into fiber shapes.
A “metal complex” refers to the state where molecules or ions are bonded regularly around metal ions, and this indicates that the organic substances or inorganic compounds show properties and reactions that cannot be expressed in isolation.
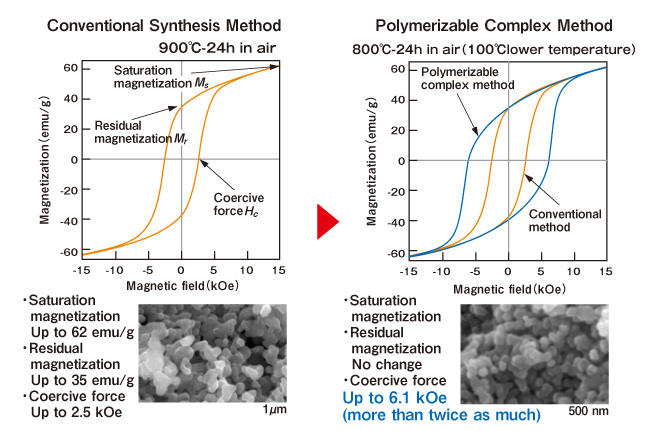
Fig. Comparison of Conventional Synthesis Method and Polymerizable Complex Method (M-type Ba ferrite)
Multiferroics: Widening Possibilities As Materials for Electronic Devices
Dr. Kikuchi’s study started from the goal of creating the ultimate ferrite as a permanent magnet material, and it has proceeded in an unexpected direction. It was reported that Sr-Co-Fe-based Z-type ferrites act as multiferroics, which combine ferromagnetic properties with ferroelectric properties. In other words, applying a magnetic field (electric field) will induce electric polarization (magnetization) of the material.
Associate professor Kikuchi explains the potential for multiferroics, “They have a wide range of applications. For example, magnetic recordings such as hard disks generate a magnetic field to perform writing, and read the magnetization to convert it to electric signals. When magnetic properties and dielectric properties affect each other, data is saved in the magnetization section, and reading and writing can be performed using an electric field and polarization. In other words, direct writing and reading of magnetic recordings can be performed electrically using an electric field and electric polarization. Also, because the voltage changes when a magnetic field is sensed, this property can be used to develop a magnetic sensor not requiring a power supply.”

Many Similarities Shared Between Superconductors and Ferrites Replacing of Elements to Control Functions
When associate professor Kikuchi was a student, interest in high-temperature superconductivity was at its peak. This also influenced Dr. Kikuchi, who was studying material synthesis, to conduct research in superconductive materials. Later, he changed over to research on ferrites at the University of Hyogo. He explained, “Superconductors are oxides of copper, and ferrites are oxides of iron, and so they share many similarities. For both, the crystal structure is layered, and they can be classified by combinations of basic structural units. Although current flows without resistance in superconductors, the manifestation of superconductivity is closely related to magnetism. It was at that time that I came across the synthesis method known as the polymerizable complex method.”
Currently, the main focus of associate professor Kikuchi’s research continues to be multiferroics. To improve the magnetization of magnets, one simply needs to align a collection of small magnets all in the same direction. Dr. Kikuchi says, “In developing multiferroics, it is clusters that are tilted with respect to the crystal axis, not those facing the same direction, that will become multiferroics. If some method can be found to change this direction, multiferroics themselves can be controlled.” Then, what is the state that brings out their optimal properties, or what method can be used for constraining and averaging out their properties? He has already succeeded using the polymerizable complex method to replace materials at the molecular level in the synthesis of key materials. The magnetic properties were also measured. The results showed ferromagnetic properties, but it is unclear whether superior ferroelectric properties were also exhibited. Many issues remain as topics for future study.
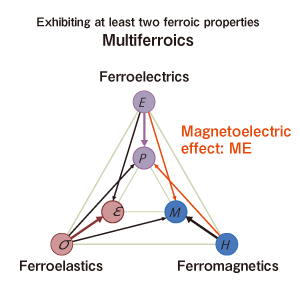
Multiferroics
The term multiferroics refers to substances having at least two of the following properties indicating a type of response to an external force: Ferroelectricity, ferromagneticity, and ferroelasticity. In the figure below, an electric polarization (P) occurs due to an electric field (E), magnetization (M) occurs due to a magnetic field (H), and strain (ε) occurs due to stress (δ). This shows that these results are based on range-dependent order. Ferroic is a concept that comprehensively describes their common properties, and multiferroic is a term formed by combining multi- and ferroic to describe how the substances exhibit multiple primary ferroic order parameters simultaneously.
Multiferroic substances are expected to yield new responses due to interaction of different ordered states. For example, instead of the responses of conventional materials, such as “magnetic field ←→ magnetization” and “electric field ←→ electric polarization,” we can obtain responses like “magnetic field ←→ electric polarization” and “electric field ←→magnetization.” This type of condition is called the magnetoelectric effect, and it used to occur only at low temperatures or when a strong magnetic field was applied, but this effect has been demonstrated for Sr-Co-Fe-based Z-type ferrites at room temperatures. For this reason, potential applications include memory, switching elements, and other devices based on these new operation principles.